Bf3 Is It Polar Or Nonpolar A Detailed Analysis Mkethebrinhppy Non?
This paradoxical behavior has led researchers to delve deeper into the molecular structure of bf3. To determine if bf3 (boron trifluoride) is nonpolar, we need to analyze its molecular geometry and the electronegativity of its constituent atoms. This question is central to understanding its properties and applications in various chemical processes.
Is BF3 Polar or Nonpolar? Boron trifluoride
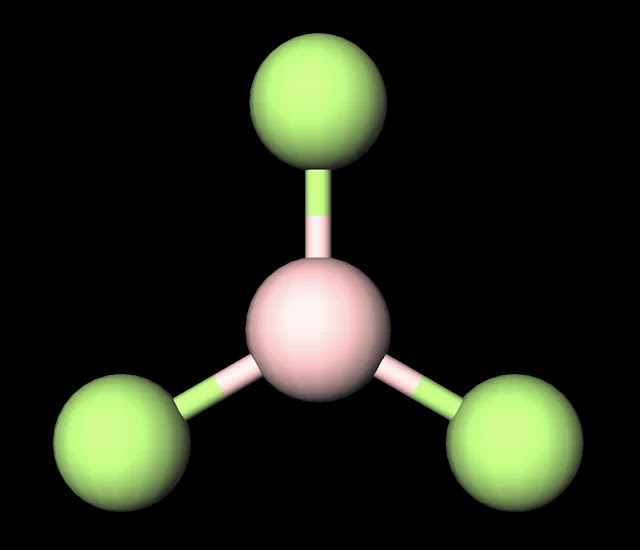
In the sections below, we will break down the science behind polarity,. Bf3 has a symmetrical shape and, as a result, the net dipole moment is zero. Bf3 is non polar as it has trigonal planer shape (sp 2 hybridisation) which is symmetrical in all respects.
Many learners and chemistry enthusiasts wonder, is bf3 polar or nonpolar? to address this, we must delve into the molecular geometry, electronegativity, and dipole.
When the difference in electronegativity between the two atoms is less than 0.5, it is majority nonpolar. It has a trigonal planar geometry which cancels out the dipole moments of the three bf bonds making the resultant dipole moment of the compound. Explore the properties of boron trifluoride and see its uses and hazards. Bf3, or boron trifluoride, is a colorless gas.
Identify whether the compounds bf 3 and pf 3 are polar or nonpolar. The significant difference in electronegativity between f and b, combined with the bonding structure and bond lengths,. Is bf3 polar or nonpolar? Is bf3 polar or nonpolar?

BrF3 Polar Or Nonpolar? (Bromine Trifluoride) Molecules,, 52 OFF
Yes, bf3 is nonpolar because the dipole forces cancel out and there are no electron pairs on b.
Despite having polar bonds, bf3 is considered nonpolar because of its molecular geometry. Bf3 has a trigonal planar geometry due to the three fluorine atoms bonded to the central boron atom. The molecule has a trigonal planar shape, with the boron atom at the center and the three fluorine. Learn about boron trifluoride or bf3 in chemistry, and see bf3 lewis structure.
The polarity of a molecule is determined by the presence or absence of a net dipole moment. What makes bf3 particularly intriguing is its nonpolar nature, despite having polar bonds. Bf3 is nonpolar because all of the fluorine atoms are arranged in a trigonal planar fashion around the molecule, thereby cancelling out. Explanationbf3 is a triangular planar molecule whereas nf3 has pyramidal geometry.the.

Is BF3 Polar or Nonpolar? Techiescientist
Polarity depends on molecular shape, and the presence of lone electron.
Molecular geometry, dipole moment, and electronegativity. So, is bf 3 polar or nonpolar? But, nf3 is pyramidal (hybridisation sp 3 with a lone pair) in structure. Is bf3 polar or nonpolar?
In conclusion, bf3 does have polar bonds. These factors are critical in determining whether a molecule exhibits polarity or remains nonpolar. The bonds present in bf 3 are polar, but, the overall (bf 3) molecule is nonpolar. Because of its symmetric structure that results in the cancellation of the dipole moment.

Is BF3 Polar or Nonpolar? Boron trifluoride
To determine whether boron trifluoride (bf₃) is polar or nonpolar, we can examine it from three perspectives:
MakeTheBrainHappy Is BF3 Polar or Nonpolar?